|
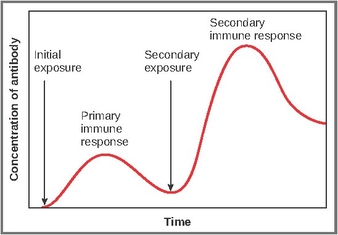
Caitlyn Tran 5.4 is greater than 0 and 0 is less than 5.4. With the basic rules of math, comparing these numbers seems so simple, yet they become more complex when applied to topics like representation. As of 2016, Asian Americans and Pacific Islanders (AAPIs) make up approximately 5.4% of the United States population. Although that does not sound like much, this mere 5.4% translates to over 16 million people. With such large variations between different ethnic groups, including but not limited to differences in socioeconomic levels and differences in culture, it makes sense that their health and medical needs may differ too and that representation in clinical trials is needed. However, this has not been the case, as seen with how the racial and ethnic minorities that make up approximately 30% of the United States population only constituted 18% of clinical trial subjects in cancer clinical trials funded by the National Cancer Institute between January 1, 2003, and June 30, 2005. This indicates that some barriers must be responsible for the discrepancy between clinical trial representation and minority representation in the United States population. Multiple studies have confirmed this by providing insight into what those barriers may be. Myths about AAPIs being a healthy minority, problems with informed consent, and mistrust in healthcare are a few that have been brought up. Clinical trial representation could potentially increase if the aforementioned barriers are addressed. For instance, healthcare professionals could play a larger role in increasing trust in healthcare by being more culturally aware and sensitive in addition to making a larger effort to recruit more AAPI members to participate in clinical trials. This topic has become even more significant in light of the distribution of two COVID-19 vaccines: one by Pfizer/BioNTech and the other by Moderna. In the past, there have been several different vaccine types, which contained different components: inactivated or weakened virus, harmless fragments of proteins or protein shells, a viral vector in which a virus is genetically engineered to generate proteins, or RNA or DNA genetically engineered to generate proteins. Regardless of the type, each vaccine has been designed to elicit a primary immune response. This way, the body can manufacture memory cells and other immune cells required to elicit a faster and more effective secondary immune response if the body ever comes across the actual pathogen. Since the COVID-19 pandemic began, the pressure to quickly produce a vaccine has encouraged scientists to explore a new type of vaccine: an mRNA vaccine, which was used by both Pfizer/BioNTech and Moderna. Once the COVID-19 vaccine is administered to muscle in the arm, muscle cells can use the mRNA to produce spike proteins found on the surface of the actual SARS-CoV-2 virus before breaking down the mRNA. The proteins still generate an immune response, but the mRNA cannot cause the virus nor does it interfere with a person’s genetic material. This makes the vaccine safe and open for everyone to take unless they have had an allergic reaction to any past vaccines or COVID-19 vaccine components. However, further details should be discussed with primary care doctors or relevant healthcare specialists. Even with these details about how the vaccine should theoretically work, the safety and efficacy of a vaccine ultimately boils down to clinical trial results, which brings us back to the matter of representation in clinical trials. As supported by evidence that clinical trials are becoming more diverse, the vaccine by Pfizer/BioNTech clinical trial racial and ethnic distribution included 26.2% Hispanics/Latinos, 9.8% African Americans, 4.4% Asians, and less than 3% other minorities. Similarly, Moderna’s vaccine clinical trials included 20% Hispanics, 9.7% African Americans, 4.7% Asians, and less than 3% all others. With so many different racial and ethnic groups present in the United States population, these clinical trials could be more diverse. However, the breakdown of the recent COVID-19 vaccines indicates that we are taking a step in the right direction. Hopefully, this trend continues and AAPI along with other minority representation in clinical trials will improve both health results and trust in healthcare. Since AAPIs represent about 5.4% of the United States population, they should also be represented in clinical trials. After all, 5.4 is greater than zero. Sources:
0 Comments
|
Archives
April 2021
Categories |